
Virendra Kumar- CEO, AdametNext
Over the last decade, India has rapidly emerged as a global hub for pharmaceutical Global Capability Centers (GCCs). These centers, which serve as critical extensions of multinational pharma companies, have transformed the pharmaceutical landscape in the country by fostering innovation, boosting employment, and contributing significantly to economic growth. Along with areas such as manufacturing and research, Pharma business support services have been accepted as a key offering by pharma GCCs. Over a period, customer expectations have shifted towards more value-oriented services. India has become the home of pharma experts who understand the critical aspects of decision-making during drug development and go-to-market (GTM) strategy planning.
Pharma GCCs are offshore units of Pharma MNCs. These GCCs are set up to centralize operations, research, and support services. These centers play a crucial role in drug discovery, regulatory compliance, pharmacovigilance, data analytics, clinical research, commercialization (brand planning, marketing, and sales), and digital transformation in the pharmaceutical sector. Pharma GCCs enable global companies to streamline their operations while leveraging their host country’s technological expertise and cost efficiency, such as the US, EU, and UK markets.
The big pharma companies initially tested the water in collaboration with various India-based KPOs such as WNS, EvalueServe, ZS, Cognizant, etc. In the early 2000s, the less critical and time-consuming business activities were shifted to India. With the knowledge transfer, more and more complex services were also moved to India, such as commercial and medical integrated insights, competitive readiness leads, commercial analytics, forecasting, and market access solutions.
With the passage of time, many Pharma MNCs initiated the establishment of these captives under their brands as done by companies like Novartis, Lilly, BMS, Sandoz, Amgen, Sanofi, and Takeda to name a few. Regarding location priority, Hyderabad was a clear winner among other cities.
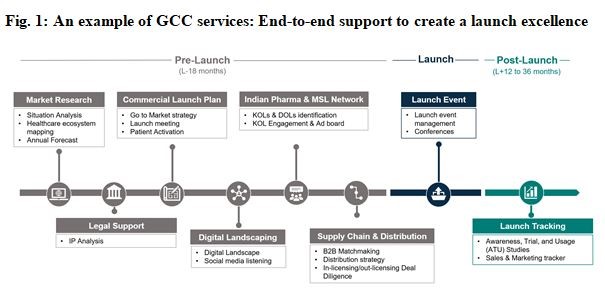
Product Life Cycle Services typically include Commercial Services, Scientific Services, and Operations Services. Insights and Analytics, Field Force Operations, and Patient Access are integral components of Commercial Services. Medical Support, Pre-Launch Scientific Support, and Post-Launch Scientific Support are integral parts of Scientific Services, whereas Tech Operations Support, Legal Compliance & Intellectual Property (IP), and Lean Six Sigma are integral components of Operations Services. Certain services are part of Commercial Services as well as Scientific Services, e.g., Customer Campaigns & Market Development. There are services such as Training Design & Delivery, which cover the entire gamut of PLC Services covering Commercial, Scientific & Operations Services.
India is becoming a preferred destination for Pharmaceutical GCCs due to multiple factors. The availability of a skilled pharmaceutical workforce is a major factor due to the vast pool of pharmaceutical graduates and postgraduates, including highly skilled professionals in MBA/M. Pharm. and other specialized fields such as clinical research and healthcare service management. With over 4 Lakh pharmaceutical graduates annually, the country offers a young, talent-rich ecosystem for pharmaceutical GCCs who can easily be trained in global pharmaceutical operation management.
India offers lower operational costs while maintaining high-quality output compared to developed nations. Companies setting up GCCs in India benefit from reduced labour and infrastructure expenses, making it an attractive investment destination. The efficiency gain helps the global companies maintain their positive bottom line while their top line performance shrinks due to poor new launch performance or blockbuster drug patient cliff.
Strong IT and digital infrastructure have brought the global workforce close to each other, and now people in the US and India can work like colleagues without many communication challenges. The acceptance of remote work has enabled work-from-anywhere concepts across the globe.
The Government of India has proactively promoted foreign investment through initiatives like Make in India, Start-up India, and production-linked incentives (PLI). Setting up Special Economic Zones (SEZs) helped provide a conducive environment for pharma GCCs.
Indian pharma professionals have traveled a long way in the last 20-25 years, thereby gaining recognition for their work and potential. Many Indian professionals have also successfully shifted to the US and EU to take bigger responsibilities in the pharma value chain. The confidence in the Indian workforce has led to the shift of more business-critical roles to India.
The rapid expansion of pharma GCCs in India has significantly impacted the economy in multiple ways, be it job creation, strengthening of the Innovation Ecosystem, FDI, or boosting ancillary industries. Along with conventional job options, offshore GCCs (captive/vendors) have emerged as major high-value job-creating centers. Due to their global exposure and best employee-friendly practices, these centers have emerged as preferred employers in recent years.
The majority of professionals in the global GCC support global innovative drug development. It equipped them with the essential skills required for converting a new chemical entity into a pharmaceutical drug/brand. Unfortunately, these skills are not much appreciated by the local pharmaceutical industry, which revolves around low-cost generic options. However, the exposure has inspired many young professionals to work in innovative drug development businesses.
The expansion of GCCs (now close to 1,700), including Pharma sector-specific GCCs, has attracted significant FDI inflows, strengthening India’s economic resilience and fostering a stable business environment. Besides attracting FDI, the presence of pharma GCCs fuels growth in associated industries such as biotechnology, software development, data security, and legal services, further expanding India’s economic potential.
To maximize the Pharma GCC opportunity, continuous efforts are required from all stakeholders. The government should continue to refine its policies to attract more foreign investments in the pharma GCC sector. Streamlining regulatory approvals, offering tax incentives, and facilitating ease of business can further solidify India’s position as a global hub. By increasing funding for R&D initiatives and fostering industry-academia collaboration, India can drive innovation in pharmaceuticals and biotech, strengthening the long-term sustainability of pharma GCCs. Investing in AI, big data analytics and automation technologies will help pharma GCCs optimize operations and develop novel drug discovery and patient care solutions. Expanding healthcare and IT infrastructure, particularly in Tier 2 and Tier 3 cities, can support the decentralization of pharma GCCs and create employment opportunities in emerging regions.
The most pertinent intervention is to align our education system with industry requirements to sustain and enhance India’s attractiveness as a pharma GCC hub. Pharma institutes (national or regional) must understand and appreciate the requirements of pharma GCCs. Accordingly, these institutes must change their syllabus to make their students more relevant to pharma GCCs. Prominent institutes have already initiated these moves, e.g., in many such courses, students are now exposed to the global drug development and commercialization process rather than restricted to only the Indian market focus.
Pharma sector-specific programs such as MBA (Pharm.) at the Department of Pharmaceutical Management, NIPER Mohali, collaboration of pharma institutions with pharma GCCs and vendors/partners such as AdametNext, IQVIA, etc. can help in aligning curriculum in line with the changing demands of pharma industry including Pharma GCCs. New-age institutes must include the novel drug development and commercialization process in their syllabus to make their students industry-ready. A pertinent example in this regard is MBA students, while working with AdametNext on live projects, develop an understanding of the Commercial Thought Process (Refer to Figure 3) behind the pharmaceutical brand building.
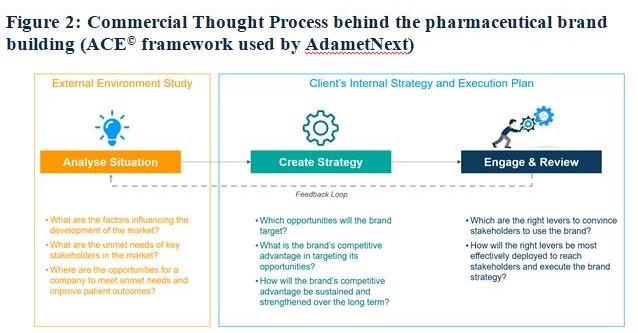
Encouraging Multidisciplinary Learning, including new-age data analytics, use of new-age tools such as Chat-GPT, and understanding of various key stakeholders in the drug development and commercialization process can ensure 360-degree readiness of Indian talent for these global GCC opportunities. Along with technical expertise, communication, critical thinking, and problem-solving skills are essential for roles in global pharma GCCs. Introducing leadership and business communication training at the academic level will help graduates integrate seamlessly into the industry.
To conclude,India’s rise as a premier destination for pharma GCCs is a testament to its robust talent pool, cost advantages, and technological capabilities. As the global pharmaceutical industry continues to evolve, India has the potential to emerge as a powerhouse for research, innovation, brand building, and commercialization of pharmaceutical brands. However, realizing this potential requires a concerted effort from policymakers, industry leaders, and educational institutions to foster a conducive environment for growth. By implementing strategic reforms and investing in future-ready talent, India can not only strengthen its position as a global pharma GCC leader but also drive long-term economic and healthcare advancements on a global scale.
Virendra Kumar- CEO, AdametNext (Hyderabad)






